Kabyashree Buragohain, Dhruba Jyoti Bordoloi and Ratul Nath*
Microbiology Lab., Dept. of Life Sciences, Dibrugarh University, Assam
*Email address of the corresponding author: ratulnath@dibru.ac.in
Abstract
Arsenic contamination in water poses a significant threat to public health and the environment, necessitating effective remediation strategies. Microbial bioremediation has emerged as a promising approach for arsenic removal, leveraging the metabolic capabilities of microorganisms to transform and immobilize arsenic species. This review presents an organized analysis of functional microorganisms involved in arsenic detoxification, highlighting their diverse metabolic pathways and mechanisms, such as arsenic oxidation, reduction, methylation, and sequestration. By integrating microbiological insights with environmental engineering strategies, this review emphasizes the promising future of arsenic bioremediation as a viable approach for mitigating arsenic toxicity and ensuring access to safe drinking water.
Keywords: Arsenic, toxicity, health hazards, arsenite, arsenate, bioremediation.
Introduction:
Arsenic (As), a toxic metalloid that ranks 20th in natural abundance, is extensively but irregularly dispersed in nature and is commonly found in rocks, soil, and water environments (Bahar et al., 2013; Yin et al., 2022). Its increasing presence in the biosphere is a matter of concern for both environmental and human health (Majumder et al., 2013), particularly because it is a persistent category 1 human carcinogen (Laha et al., 2022). The various forms of As found in natural environments and biological systems include elemental arsenic (As 0), arsine (As -3), arsenite (As III), and arsenate (As V) (Yin et al., 2022). Generally, As III is dominant in reducing environments, while As V is prevalent in oxidizing conditions (Yamamura and Amachi, 2014). Under neutral conditions, As V predominantly exists as negatively charged oxygen anions (H2AsO4- and H2AsO42-), whereas As III exists as an uncharged compound (H3AsO3) (Yin et al., 2022). Arsenic is mainly introduced into the environment via natural processes or human activities. While the typical concentration of As in groundwater is usually below 10 μg L−1 in most countries, literature reports a wide range of values from less than 0.5 to 5,000 μg L−1 occurring under natural conditions (Bahar et al., 2013). Arsenic contamination primarily stems from mining, waste discharge, fertilizer application, pesticide usage, and burning fuels (Moreno-Jiménez et al., 2012; Yin et al., 2022). Being a potential toxin, it can be liberated through rock erosion, volcanic activities, and wildfires (Verma et al., 2018). The prevalence of As is clear as it is found in over 200 minerals within the Earth’s crust. Among these, 60% exist as arsenates, 20% as sulpho-salts and sulphides, and the remaining 20% are in various forms including arsenides, arsenites, oxides, silicates, and elemental arsenic (Abbas et al., 2018).
Exposure to Arsenic has been linked to a variety of health risks, including skin and neurological issues, as well as various types of cancer. Moreover, recent findings indicate its correlation with non-communicable conditions like diabetes mellitus and cardiovascular ailments such as hypertension (Rahaman et al., 2021). As a result, the US Environmental Protection Agency (US-EPA), the International Agency for Research on Cancer, and the World Health Organization (WHO) have established a considerable limit of 10 ppb for arsenic in drinking water (Irshad et al., 2021). For many years, it has been seen that the public’s awareness of the economic and health risks associated with Arsenic contamination has increased, leading to the advancement of technologies aimed at mitigating or eliminating the contamination (Lim et al., 2014; Yin et al., 2022). Considering the toxicity of Arsenic, it has become a global concern, making it crucial to develop remediation strategies for As-contaminated water and soil (Hassan et al., 2009; Ahmad et al., 2020). In this context, a wide range of physicochemical techniques, including adsorption, chemical precipitation, co-precipitation, electroplating, ion exchange, filtration, and reverse osmosis are extensively employed for arsenic decontamination (Irshad et al., 2021). However, many of these techniques face practical challenges, such as the production of toxic sludge, inefficiency in natural settings, high operational and maintenance costs, and operational difficulties (Dwivedi and Srivastava, 2015; Irshad et al., 2021).
Conversely, bioremediation does not have any of the previously mentioned drawbacks and stands out as the optimal solution for removing arsenic from the environment (Rahman and Singh, 2020; Irshad et al., 2021). Given its remarkable effectiveness, environmental friendliness, and cost-efficiency, using microorganisms to clean up arsenic contamination is seen as a promising and innovative approach (Yamamura and Amachi, 2014; Valls and de Lorenzo, 2002; Hare et al., 2020; Yin et al., 2022). In the past few years, it has been demonstrated that bacteria linked to plants also have a significant impact on the mobilization of Arsenic. According to a recent study conducted by Ma et al. (2016), the interaction between plants and microorganisms, particularly those in the rhizosphere and the endophytic bacteria, could potentially improve the process of phytoremediation for soil and shallow groundwater contaminated with arsenic. Many microbes help their host plants by reducing the harmful effects of arsenic through their metal resistance mechanisms and by promoting plant growth (Crognale et al., 2017).
This review paper examines Arsenic contamination of water, focusing on its public health implications. It outlines the sources of Arsenic, mechanisms of toxicity, associated human diseases, and viable remediation techniques for addressing Arsenic toxicity. It demonstrates a thorough and organized understanding of the functional microorganisms involved in Arsenic remediation and their various mechanisms. It offers promising potential for the future application of Arsenic bioremediation.
Sources and toxicity of Arsenic:
Arsenic is widespread in the natural environment, making up approximately 0.00005% of the Earth’s crust, and is the 12th most abundant element in the human body (Mandal and Suzuki, 2002). The estimated global average Arsenic concentration is 5 mg/kg in soil, 1–2 μg/l in open seawater, and below 10 μg/l in portable ground and surface water. Numerous countries, such as Bangladesh, India, China, Pakistan, Chile, Argentina, Mexico, Poland, New Zealand, Canada, Hungary, Taiwan, the United States and Japan, are encountering the problem of groundwater contamination by Arsenic (Abbas et al., 2018). The occurrence of Arsenic in natural water is typically linked to geochemical environments, including basin-fill deposits from alluvial and lacustrine sources, volcanic deposits, geothermal inputs, and contamination from mining waste and landfills (Raju, 2022). Arsenic exists in the environment due to both anthropogenic and geogenic occurrences. Processes like soil formation, dust storms, volcanic eruptions, geothermal/hydrothermal activity, and forest fires contribute to the presence of Arsenic in the environment (Garelick et al., 2008; Rathi and Kumar, 2021) (Fig-1). Geological formations, such as sedimentary rocks, volcanic rocks, and soils, represent the primary natural reservoirs where As tends to accumulate (Rahaman et al., 2021; Shaji et al., 2021). Arsenic is naturally present across various environmental areas (EFSA, 2009), and is found in over 245 different minerals (Cox, 1995). In nature, only a fraction of Arsenic exists in its pure elemental state, with the majority of inorganic Arsenic typically bonded with other elements like oxygen, sulphur, and chlorine (Bundschuh et al., 2021). Iron, silver, lead, copper, nickel, antimony, and cobalt deposits are rich in minerals containing Arsenic (WHO, 2001: Bundschuh et al., 2021). Over 200 minerals contain Arsenic, which is found in crustal materials. The primary sources of groundwater contamination among these are Realgar (As4 S4), Arsenopyrite (FeAsS), Anargite (Cu3 AsS4), Arsenolite, and Orpiment. Other notable minerals include Loellingite, Safforlite, Niccolite, Rammelsbergite, Cobaltite, Enargite, Gerdsorfite and Glaucodot (Madhukar et al., 2016).
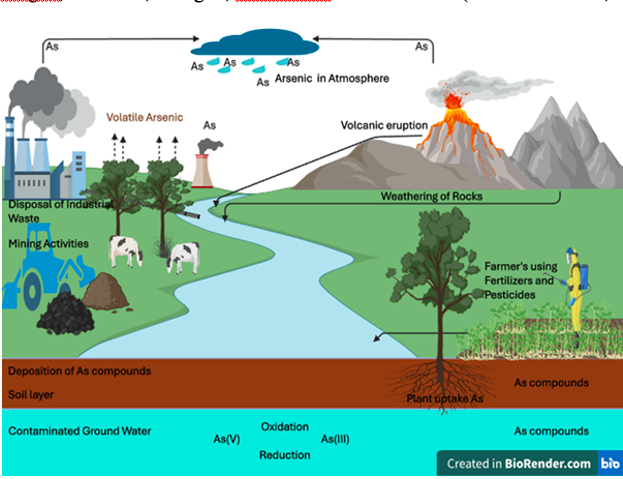
Fig. 1. Sources of Arsenic contamination
The primary human activities that contribute to groundwater contamination with Arsenic include mining, fossil fuel combustion, agricultural use of As-based fungicides, herbicides, and insecticides, as well as the application of wood preservatives (Nriagu et al., 2007). Burning coal significantly contributes to Arsenic contamination in the environment. During coal combustion, Arsenic is emitted to the atmosphere as arsenic trioxide (As4O6). This compound condenses in the flue gas system and eventually ends up in water bodies (Bissen and Frimmel, 2003). The level of groundwater Arsenic contamination from the previously mentioned human activities is significantly lower than that from natural sources, yet their impact should not be overlooked (Shankar et al., 2014).
Due to its widespread presence, toxicity, and cancer-causing properties, Arsenic is regarded as one of the most dangerous chemicals in the world (Rahaman et al., 2021). Over one hundred million individuals are at significant risk of increased Arsenic exposure, primarily through drinking water and also from industrial emissions (Prakash and Verma, 2020). The presence of Arsenic in drinking water leads to toxic and carcinogenic effects in humans. Arsenic is the first metalloid recognized as a human carcinogen, and most cases of chronic arsenicosis result from the prolonged consumption of As-contaminated water (Prakash and Verma, 2021). The toxicity of Arsenic is significantly influenced by its physical state and the chemical form of the Arsenic compound. Arsenic toxicity can lead to bladder, prostate, lung, and skin cancers, as well as rhagades, skin lesions, oxidative stress, mitochondrial damage, and potential disruption of DNA methylation or DNA repair systems (Satyapal and Kumar, 2021).
Prolonged exposure to inorganic arsenic (iAs) impacts the skin, especially in areas where it folds. This exposure can lead to changes in pigmentation, hyperkeratosis, and eventually, the development of skin cancer, such as on the hands. Arsenic is a well-documented human carcinogen that affects multiple organs (Nurchi et al., 2020). Significantly higher risks of lung, urinary tract, and skin cancers have been reported when drinking water contains As levels around or above 50 µg/L (National Research Council, 2001). Studies examining exposure to Arsenic in various populations have indicated a potential slight increase in the likelihood of developing bladder cancer even at Arsenic concentrations in drinking water as low as 10 µg/L (Mink et al., 2008), despite a recent meta-analysis indicating no heightened risk at this threshold (Boffetta and Borron, 2019; Nurchi et al., 2020).
Conventional methods for removal of Arsenic
Over recent years, there has been considerable research conducted on methods for removing Arsenic to enhance its extraction from the environment. Scientists are scrutinizing these techniques through both laboratory trials and field studies. The techniques presently employed include ion exchange, adsorption, chemical precipitation, electrocoagulation, and membrane processes (Alka et al., 2021). Although these processes are effective in eliminating Arsenic from soil and water, each innovation has notable limitations, especially regarding costs and efficiency.
Ion exchange:
Ion exchange is a physicochemical treatment process used to eliminate Arsenic or other pollutants. Ions held electrostatically on the surface of a solid phase can be replaced by ions of the same charge from the solution (Katsoyiannis and Zouboulis, 2006). This efficient technique is mainly employed to soften water and remove contaminants like arsenate, selenite, nitrate, and chromate anions from polluted water or wastewater. The process involves the use of routinely regenerated resins through which the contaminated water flows, allowing the exchanged ions to be recycled and strengthened (Al-Jubouri and Holmes, 2020; Alka et al., 2021). Polymeric and inorganic ion-exchangers, including chitosan and zeolite-based types, have been utilized as foundations for creating modified ion exchangers. The chemical and physical properties of the host materials, such as pore size and distribution, are crucial for improving the removal efficiency of the final product. Consequently, modified ion-exchange has proven to be an effective technique for the selective extraction of Arsenic species from water (Weerasundara et al., 2021).
Adsorption:
Adsorption is a widely used technique in studies focused on the selective removal of Arsenic. Iron-based methods are particularly prevalent for achieving selective Arsenic removal through adsorption (Weerasundara et al., 2021). However, not all adsorptive materials are the same. Hlavay & Polyák (2005) developed an innovative adsorptive material for removing As III and As V from water by precipitating Fe(OH)3 in situ on the surface of granulated activated Al2O3, which serves as the supporting material (Weerasundara et al., 2021).
Chemical precipitation:
Chemical precipitation involves using reagents such as ferric salts, sulphides, magnesium salts, and calcium salts to remove heavy metals like Arsenic. These agents aid in the removal of Arsenic from wastewater by converting dissolved Arsenic into low-solubility compounds, and they are also applicable in solid waste stabilization. The most prevalent methods for precipitating Arsenic in wastewater include the formation of calcium arsenate and ferric arsenate. However, calcium arsenate can become unstable in the presence of water and carbon dioxide, transforming into calcium carbonate and arsenic acid. Ferric arsenate, on the other hand, varies in stability, with amorphous ferric arsenate being less stable and crystalline ferric arsenate being more stable under specific conditions (Long et al., 2019; Alka et al., 2021).
Electrocoagulation:
The electrocoagulation method has been recognized as one of the simplest and most effective techniques for selective Arsenic removal (Ali et al., 2013). In this method, the fundamental process for eliminating As III from water consists of two steps: The initial step involves transforming As III into As V. Because As III converts to As V, the removal process is typically slowed by the presence of As III (Kumar et al., 2004; Wan et al., 2011; Weerasundara et al., 2021). The second step involves the removal of As V by generating coagulants through the oxidation of the sacrificial anode. This process is divided into three sub-steps. Initially, when voltage is applied, the sacrificial anode undergoes oxidation, producing a coagulant that is insoluble in water. Common anodes used in electrocoagulation for Arsenic removal include zinc (Zn), iron (Fe), and copper (Cu) ( Wan et al., 2011; Ali et al., 2013; Weerasundara et al., 2021).
Membrane processes:
Membrane-based techniques like reverse osmosis are cost-effective for water containing only trace amounts of contaminants. Generally, Arsenic contamination is found at trace levels (below 500 μg/L), making reverse osmosis a viable method for Arsenic removal in most cases (Sarkar et al., 2012). The reverse-osmosis technique for Arsenic removal has been recognized as very effective. However, it demands high operating pressure and significant energy, which raises the process costs and decreases its overall efficiency (Chang et al., 2014; Fox et al., 2016). Forward osmosis operates at low pressure and exhibits minimal fouling tendencies, which can consequently lower the costs associated with the removal process (Weerasundara et al., 2021).
Bioremediation of Arsenic
Microorganisms are found everywhere, spanning diverse habitats, and they hold significance in maintaining ecological equilibrium and promoting human well-being. Through altering the valence states of Arsenic, microorganisms mitigate its toxicity, consequently influencing its movement and alteration within ecosystems, either directly or indirectly (Yamamura et al., 2003; Sher and Rehman, 2019). The application of microorganisms is gaining growing interest in various industries like food and beverages, cosmetics, pharmaceuticals, and energy (Weerasundara et al., 2021). In contrast to the organic pollutants which naturally break down, Arsenic doesn’t degrade on its own. However, it can be eliminated and made less harmful by altering its solubility and state with the help of microorganisms (Rahman and Singh, 2020). Over the past decade, research has shown that a wide range of microorganisms, such as bacteria, fungi, and archaea, can facilitate the process of bioremediation to tackle Arsenic contamination (Hare et al., 2020; Yin et al., 2022). Nevertheless, utilizing microorganisms for Arsenic removal from water and wastewater remains a relatively new approach (Weerasundara et al., 2021).
Bacterial uptake of arsenate and arsenite primarily relies on the phosphate transporter and aquaglycoporins, respectively. Therefore, bacteria do not need to develop specialized mechanisms for Arsenic uptake. Additionally, some bacteria possess genetic mechanisms to resist both organic and inorganic arsenic. The widely recognized operon for bacterial Arsenic tolerance and detoxification is the ars operon, which reduces arsenate to arsenite for detoxification (Kruger et al., 2013). Additionally, bacteria oxidize Arsenic species through the aioA and aioB genes, which encode arsenite oxidase, the crucial enzyme for converting arsenite to arsenate (Kabiraj et al., 2022). In the bacterial ars operon, the ArsC enzyme is crucial for arsenate reduction and contributes to Arsenic resistance, as previously described. Bacterial Arsenic transformation includes respiratory As V reduction and As III oxidation, which support autotrophic growth and is collectively known as “arsenotrophy.” (Oremland et al., 2009). In arsenotrophy, the conversion between As V and As III is facilitated by one of three enzymes from the dimethyl sulfoxide (DMSO) reductase family: arsenate reductase (ARR) and two distantly related enzymes, arsenite oxidases AIO and ARX (Ospino et al., 2019).
Researchers globally have been consistently studying arsenic-resistant bacteria, with a particular focus on understanding their molecular characteristics. The regulation of Arsenic tolerance is precise, but the degree of tolerance differs between species and even among strains (Kabiraj et al., 2022). Bacteria possessing Arsenic tolerance include, Staphylococcus, Escherichia, Achromobacter, Fucus gardneri, Sinorhizobium, Rhodobium, Pseudomonas, Bradyrhizobium, Cupriavidus, Clostridium, Agrobacterium, Thiobacillus, Psychrobacter species, Acinetobacter, Microbacterium, Rhodococcus, Penicillium gladioli, Alcaligens, Desulfomicrobium, Ochrobactrum, Bosea species, Fomitopsis pinicola, Fusarium oxysporum, and Rhodobium (Chandra and Banik, 2021; Laha et al., 2022).
Biofilm formation has also been noted among these PGPRs. Typically, the biofilm structure consists of EPS, proteins, and extracellular nucleic acids (Armendariz et al., 2015) and (Vezza et al., 2020) observed that it forms boundaries around cell populations on its surface, which further hinders metal diffusion. The most extensively studied approach remains the active extrusion of metal from the bacterial cell. Extrusion facilitated by the ars operon (ArsB/ArsAB) is widely distributed and well-characterized (Mandal et al., 2021). Sinorhizobium meliloti has a unique detoxification pathway where As III is expelled through an aquaglyceroporin encoded by AqpS. Interestingly, AqpS takes the place of arsB within the ars operon (Yang et al., 2012; Laha et al., 2022).
Future perspectives
Future development in microbial remediation of arsenic-contaminated water will rely on interdisciplinary research integrating microbiology, environmental engineering, chemistry, and policy development. Enhancing microbial arsenic transformation through genetic engineering and synthetic biology can improve detoxification efficiency. Understanding microbial interactions in complex environments will help optimize remediation strategies, while bioaugmentation and biostimulation approaches can enhance microbial activity in situ. Integrating microbial remediation with other treatment technologies, such as adsorption and filtration, can improve removal efficiency. However, large-scale implementation requires pilot studies to assess feasibility and long-term stability under real-world conditions. Environmental factors, including climate change, must also be considered to ensure adaptability and resilience of microbial remediation systems. Additionally, regulatory frameworks should be established to facilitate the adoption of these technologies, and public awareness initiatives should engage local communities for successful implementation. Addressing these challenges will pave the way for microbial bioremediation to become a mainstream solution for mitigating arsenic pollution and ensuring access to safe drinking water worldwide.
Discussion and Conclusion
Bacterial remediation in mitigating Arsenic contamination to purify water has demonstrated promising potential for tackling the widespread problem of Arsenic pollution. Conventional approaches, although successful, frequently entail disadvantages like steep expenses, chemical utilization, and the creation of additional waste. Bioremediation presents a more environmental-friendly option, harnessing the innate capacities of microorganisms to cleanse arsenic-polluted surroundings. Recent advancements have concentrated on improving the effectiveness and versatility of bioremediation methods. These alterations empower the microorganisms to flourish in polluted surroundings and convert Arsenic into less harmful forms more effectively. Moreover, biofilm-based strategies have demonstrated potential because of their large surface area and protective structures, which boost microbial durability and performance across diverse environmental settings. The accessibility of Arsenic in various water compositions, microbial resilience, and the influence of environmental variables like pH, temperature and the existence of additional pollutants can affect the efficiency of bioremediation. Furthermore, collaborative knowledge from microbiology, biotechnology, environmental science, and engineering has the potential to create strategies that tackle the technical and the ecological dimensions of Arsenic bioremediation. Cooperation among academia, industry and policymakers will be crucial in transforming small-scale laboratory advancements into feasible, large-scale remedies.
Arsenic bioremediation stands as an encouraging frontier in the pursuit of efficient and eco-friendly water purification techniques. Recent progress, especially in genetic manipulation, the application of biofilms, and nanobiotechnology, has greatly boosted the effectiveness of bioremediation methods. Although challenges such as environmental fluctuations, microbial resilience, and biosafety issues persist, the outlook for arsenic bioremediation appears optimistic. To unlock the complete benefits of bioremediation, continuous investigation should prioritize enhancing microbial efficiency, guaranteeing environmental security, and creating adaptable applications. Prospective avenues involve refining genetically altered organisms, discovering new microbial combinations, and merging bioremediation with other water purification methods. By tackling these elements, we can progress towards more efficient and eco-friendly approaches to combat Arsenic contamination, thereby safeguarding public health and conserving essential water reservoirs.
References:
Abbas, G., Murtaza, B., Bibi, I., Shahid, M., Niazi, N. K., Khan, M. I., Amjad, M., Hussain, M., & Natasha. (2018). Arsenic Uptake, Toxicity, Detoxification, and Speciation in Plants: Physiological, Biochemical, and Molecular Aspects. International Journal of Environmental Research and Public Health, 15(1): Article 1. https://doi.org/ 10.3390/ijerph15010059
Ahmad, I., Tahir, M., Daraz, U., Ditta, A., Hussain, M. B., & Khan, Z. U. H. (2020). Responses and Tolerance of Cereal Crops to Metal and Metalloid Toxicity. In: M. Hasanuzzaman (Ed.), Agronomic Crops: Volume 3: Stress Responses and Tolerance (pp. 235–264). Springer. https://doi.org/10.1007/978-981-15-0025-1_14
Ali, I., Asim, Mohd., & Khan, T. A. (2013). Arsenite removal from water by electro-coagulation on zinc–zinc and copper–copper electrodes. International Journal of Environmental Science and Technology, 10(2): 377–384. https://doi.org/ 10.1007/s13762-012-0113-z
Al-Jubouri, S. M., & Holmes, S. M. (2020). Immobilization of cobalt ions using hierarchically porous 4A zeolite-based carbon composites: Ion-exchange and solidification. Journal of Water Process Engineering, 33: 101059. https://doi.org/ 10.1016/j.jwpe.2019.101059
Alka, S., Shahir, S., Ibrahim, N., Ndejiko, M. J., Vo, D.-V. N., & Manan, F. A. (2021). Arsenic removal technologies and future trends: A mini review. Journal of Cleaner Production, 278: 123805. https://doi.org/10.1016/j.jclepro.2020.123805
Armendariz, A. L., Talano, M. A., Wevar Oller, A. L., Medina, M. I., & Agostini, E. (2015). Effect of arsenic on tolerance mechanisms of two plant growth-promoting bacteria used as biological inoculants. Journal of Environmental Sciences, 33: 203–210. https://doi.org/10.1016/j.jes.2014.12.024
Bahar, M. M., Megharaj, M., & Naidu, R. (2013). Bioremediation of Arsenic-Contaminated Water: Recent Advances and Future Prospects. Water, Air, & Soil Pollution, 224(12): 1722. https://doi.org/10.1007/s11270-013-1722-y
Bissen, M., & Frimmel, F. H. (2003). Arsenic — a Review. Part I: Occurrence, Toxicity, Speciation, Mobility. Acta Hydrochimica et Hydrobiologica, 31(1): 9–18. https://doi.org/10.1002/aheh.200390025
Boffetta, P., & Borron, C. (2019). Low-Level Exposure to Arsenic in Drinking Water and Risk of Lung and Bladder Cancer: A Systematic Review and Dose–Response Meta-Analysis. Dose-Response, 17(3): 1559325819863634. https://doi.org/10.1177/ 1559325819863634
Bundschuh, J., Schneider, J., Alam, M. A., Niazi, N. K., Herath, I., Parvez, F., Tomaszewska, B., Guilherme, L. R. G., Maity, J. P., López, D. L., Cirelli, A. F., Pérez-Carrera, A., Morales-Simfors, N., Alarcón-Herrera, M. T., Baisch, P., Mohan, D., & Mukherjee, A. (2021). Seven potential sources of arsenic pollution in Latin America and their environmental and health impacts. Science of The Total Environment, 780: 146274. https://doi.org/10.1016/j.scitotenv.2021.146274
Chandra, R., & Banik, A. (2021). Chapter 16—Detoxification and bioconversion of arsenic and chromium. In S. Ghosh & T. J. Webster (Eds.), Nanobiotechnology (pp. 253–270). Elsevier. https://doi.org/10.1016/B978-0-12-822878-4.00016-X
Chang, F., Liu, W., & Wang, X. (2014). Comparison of polyamide nanofiltration and low-pressure reverse osmosis membranes on As III rejection under various operational conditions. Desalination, 334(1): 10–16. https://doi.org/10.1016/j.desal.2013.11.002
Cox, M. (1995). Arsenic Characterization in Soil and Arsenic Effects on Canola Growth. LSU Historical Dissertations and Theses. https://doi.org/10.31390/ gradschool_disstheses .6003
Crognale, S., Amalfitano, S., Casentini, B., Fazi, S., Petruccioli, M., & Rossetti, S. (2017). Arsenic-related microorganisms in groundwater: A review on distribution, metabolic activities and potential use in arsenic removal processes. Reviews in Environmental Science and Bio/Technology, 16(4): 647–665. https://doi.org/10.1007/s11157-017-9448-8
Dwivedi, A., & Srivastava, S. (2015). Biological Wastes the Tool for Biosorption of Arsenic. Bioremediation & Biodegradation ISSN: 2155-6199, 7: 323. https://doi.org/ 10.4172/2155-6199.1000323
Fox, D. I., Stebbins, D. M., & Alcantar, N. A. (2016). Combining Ferric Salt and Cactus Mucilage for Arsenic Removal from Water. Environmental Science & Technology, 50(5): 2507–2513. https://doi.org/10.1021/acs.est.5b04145
Garelick, H., Jones, H., Dybowska, A., & Valsami-Jones, E. (2008). Arsenic Pollution Sources. In: Reviews of Environmental Contamination, Volume 197: International Perspectives on Arsenic Pollution and Remediation (pp. 17–60). Springer. https://doi.org/10.1007/978-0-387-79284-2_2
Guidelines_for_DRINKING_WATERQUALITY.pdf. (n.d.).
Hare, V., Chowdhary, P., & Singh, A. K. (2020). 4 – Arsenic toxicity: Adverse effect and recent advance in microbes mediated bioremediation. In: P. Chowdhary, A. Raj, D. Verma, & Y. Akhter (Eds.), Microorganisms for Sustainable Environment and Health (pp. 53–80). Elsevier. https://doi.org/10.1016/B978-0-12-819001-2.00004-8
Hassan, K. M., Fukuhara, T., Hai, F. I., Bari, Q. H., & Islam, Kh. Md. S. (2009). Development of a bio-physicochemical technique for arsenic removal from groundwater. Desalination, 249(1): 224–229. https://doi.org/10.1016/ j.desal.2008.08.015
Hlavay, J., & Polyák, K. (2005). Determination of surface properties of iron hydroxide-coated alumina adsorbent prepared for removal of arsenic from drinking water. Journal of Colloid and Interface Science, 284(1): 71–77. https://doi.org/10.1016 /j.jcis.2004.10.032
Irshad, S., Xie, Z., Mehmood, S., Nawaz, A., Ditta, A., & Mahmood, Q. (2021). Insights into conventional and recent technologies for arsenic bioremediation: A systematic review. Environmental Science and Pollution Research, 28(15): 18870–18892. https://doi.org/10.1007/s11356-021-12487-8
Kabiraj, A., Biswas, R., Halder, U., & Bandopadhyay, R. (2022). Bacterial Arsenic Metabolism and Its Role in Arsenic Bioremediation. Current Microbiology, 79(5): 131. https://doi.org/10.1007/s00284-022-02810-y
Katsoyiannis, I. A., & Zouboulis, A. I. (2006). Comparative evaluation of conventional and alternative methods for the removal of arsenic from contaminated groundwaters. Reviews on Environmental Health, 21(1): 25–41. Scopus. https://doi.org/10.1515/ REVEH.2006.21.1.25
Kruger, M. C., Bertin, P. N., Heipieper, H. J., & Arsène-Ploetze, F. (2013). Bacterial metabolism of environmental arsenic—Mechanisms and biotechnological applications. Applied Microbiology and Biotechnology, 97(9): 3827–3841. https://doi.org/10.1007/s00253-013-4838-5
Kumar, P. R., Chaudhari, S., Khilar, K. C., & Mahajan, S. P. (2004). Removal of arsenic from water by electrocoagulation. Chemosphere, 55(9): 1245–1252. Scopus. https://doi.org/10.1016/j.chemosphere.2003.12.025
Laha, A., Sengupta, S., Bhattacharya, P., Mandal, J., Bhattacharyya, S., & Bhattacharyya, K. (2022). Recent advances in the bioremediation of arsenic-contaminated soils: A mini review. World Journal of Microbiology and Biotechnology, 38(11): 189. https://doi.org/10.1007/s11274-022-03375-5
Lim, K. T., Shukor, M. Y., & Wasoh, H. (2014). Physical, Chemical, and Biological Methods for the Removal of Arsenic Compounds. BioMed Research International, 2014: 503784. https://doi.org/10.1155/2014/503784
Long, H., Zheng, Y., Peng, Y., Jin, G., Deng, W., & Zhang, S. (2019). Comparison of arsenic(V) removal with different lead-containing substances and process optimization in aqueous chloride solution. Hydrometallurgy, 183: 199–206. https://doi.org/10.1016/j.hydromet.2018.12.006
Madhukar, M.; Sadashiva Murthy, B.M. and Udayashankara, T.H. (2016). Sources of Arsenic in Groundwater and its health significance- a review. Nature Environment and Pollution Technology, 15: 971–979.
Ma, Y., Rajkumar, M., Zhang, C., & Freitas, H. (2016). Beneficial role of bacterial endophytes in heavy metal phytoremediation. Journal of Environmental Management, 174: 14–25. https://doi.org/10.1016/j.jenvman.2016.02.047
Majumder, A., Bhattacharyya, K., Bhattacharyya, S., & Kole, S. C. (2013). Arsenic-tolerant, arsenite-oxidising bacterial strains in the contaminated soils of West Bengal, India. Science of The Total Environment, 463–464: 1006–1014. https://doi.org/ 10.1016/j.scitotenv.2013.06.068
Mandal, B. K., & Suzuki, K. T. (2002). Arsenic round the world: A review. Talanta, 58(1): 201–235. https://doi.org/10.1016/S0039-9140(02)00268-0
Mandal, J., Sengupta, S., Sarkar, S., Mukherjee, A., Wood, M. D., Hutchinson, S. M., & Mondal, D. (2021). Meta-Analysis Enables Prediction of the Maximum Permissible Arsenic Concentration in Asian Paddy Soil. Frontiers in Environmental Science, 9. https://doi.org/10.3389/fenvs.2021.760125
Mink, P. J., Alexander, D. D., Barraj, L. M., Kelsh, M. A., & Tsuji, J. S. (2008). Low-level arsenic exposure in drinking water and bladder cancer: A review and meta-analysis. Regulatory Toxicology and Pharmacology, 52(3): 299–310. https://doi.org/10.1016 /j.yrtph.2008.08.010
Moreno-Jiménez, E., Esteban, E., & Peñalosa, J. M. (2012). The Fate of Arsenic in Soil-Plant Systems. In: D. M. Whitacre (Ed.), Reviews of Environmental Contamination and Toxicology (pp. 1–37). Springer. https://doi.org/10.1007/978-1-4614-1463-6_1
National Research Council (US) Subcommittee to Update the 1999 Arsenic in Drinking Water Report. (2001). Arsenic in Drinking Water: 2001 Update. National Academies Press (US). http://www.ncbi.nlm.nih.gov/books/NBK223678/
Nriagu, J. O., Bhattacharya, P., Mukherjee, A. B., Bundschuh, J., Zevenhoven, R., & Loeppert, R. H. (2007). Arsenic in soil and groundwater: An overview. In: Trace Metals and other Contaminants in the Environment (Vol. 9, pp. 3–60). Elsevier. https://doi.org/10.1016/S1875-1121(06)09001-8
Nurchi, V. M., Buha Djordjevic, A., Crisponi, G., Alexander, J., Bjørklund, G., & Aaseth, J. (2020). Arsenic Toxicity: Molecular Targets and Therapeutic Agents. Biomolecules, 10(2): Article 2. https://doi.org/10.3390/biom10020235
Oremland, R. S., Saltikov, C. W., Wolfe-Simon, F., & Stolz, J. F. (2009). Arsenic in the Evolution of Earth and Extraterrestrial Ecosystems. Geomicrobiology Journal, 26(7): 522–536. https://doi.org/10.1080/01490450903102525
Ospino, M. C., Kojima, H., & Fukui, M. (2019). Arsenite Oxidation by a Newly Isolated Betaproteobacterium Possessing arx Genes and Diversity of the arx Gene Cluster in Bacterial Genomes. Frontiers in Microbiology, 10. https://doi.org/10.3389/ fmicb.2019.01210
Prakash, S., & Verma, A. K. (2020). Impact of Arsenic on Protein Metabolism of a Fresh Water Cat Fish, Mystus vittatus. Uttar Pradesh Journal of Zoology, 41(5): 16-19. DOI: 10.13140/RG.2.2.14789.88803
Prakash, S., & Verma, A. K. (2021). Arsenic: It’s Toxicity and Impact on Human Health (SSRN Scholarly Paper 3762024). https://papers.ssrn.com/abstract=3762024
Rahaman, Md. S., Rahman, Md. M., Mise, N., Sikder, Md. T., Ichihara, G., Uddin, Md. K., Kurasaki, M., & Ichihara, S. (2021). Environmental arsenic exposure and its contribution to human diseases, toxicity mechanism and management. Environmental Pollution, 289: 117940. https://doi.org/10.1016/j.envpol.2021.117940
Rahman, Z., & Singh, V. P. (2020). Bioremediation of toxic heavy metals (THMs) contaminated sites: Concepts, applications and challenges. Environmental Science and Pollution Research, 27(22): 27563–27581. https://doi.org/10.1007/s11356-020-08903-0
Raju, N. J. (2022). Arsenic in the geo-environment: A review of sources, geochemical processes, toxicity and removal technologies. Environmental Research, 203: 111782. https://doi.org/10.1016/j.envres.2021.111782
Rathi, B. S., & Kumar, P. S. (2021). A review on sources, identification and treatment strategies for the removal of toxic Arsenic from water system. Journal of Hazardous Materials, 418: 126299. https://doi.org/10.1016/j.jhazmat.2021.126299
Sarkar, S., Greenleaf, J. E., Gupta, A., Uy, D., & SenGupta, A. K. (2012). Sustainable Engineered Processes to Mitigate the Global Arsenic Crisis in Drinking Water: Challenges and Progress. Annual Review of Chemical and Biomolecular Engineering, 3: 497–517. https://doi.org/10.1146/annurev-chembioeng-062011-081101
Satyapal, G. K., & Kumar, N. (2021). Arsenic: Source, Distribution, Toxicity and Bioremediation. In: N. Kumar (Ed.), Arsenic Toxicity: Challenges and Solutions (pp. 153–163). Springer. https://doi.org/10.1007/978-981-33-6068-6_6
Shaji, E., Santosh, M., Sarath, K. V., Prakash, P., Deepchand, V., & Divya, B. V. (2021). Arsenic contamination of groundwater: A global synopsis with focus on the Indian Peninsula. Geoscience Frontiers, 12(3): 101079. https://doi.org/10.1016 /j.gsf.2020.08.015
Shankar, S., Shanker, U., & Shikha, U. (2014). Arsenic Contamination of Groundwater: A Review of Sources, Prevalence, Health Risks, and Strategies for Mitigation. The Scientific World Journal, 2014: e304524. https://doi.org/10.1155/2014/304524
Sher, S., & Rehman, A. (2019). Use of heavy metals resistant bacteria—A strategy for arsenic bioremediation. Applied Microbiology and Biotechnology, 103(15): 6007–6021. https://doi.org/10.1007/s00253-019-09933-6
Valls, M., & de Lorenzo, V. (2002). Exploiting the genetic and biochemical capacities of bacteria for the remediation of heavy metal pollution. FEMS Microbiology Reviews, 26(4): 327–338. https://doi.org/10.1111/j.1574-6976.2002.tb00618.x
Verma, S., Verma, P. K., Meher, A. K., Bansiwal, A. K., Tripathi, R. D., & Chakrabarty, D. (2018). A novel fungal arsenic methyltransferase, WaarsM reduces grain arsenic accumulation in transgenic rice (Oryza sativa L.). Journal of Hazardous Materials, 344: 626–634. https://doi.org/10.1016/j.jhazmat.2017.10.037
Vezza, M. E., Olmos Nicotra, M. F., Agostini, E., & Talano, M. A. (2020). Biochemical and molecular characterization of arsenic response from Azospirillum brasilense Cd, a bacterial strain used as plant inoculant. Environmental Science and Pollution Research, 27(2): 2287–2300. https://doi.org/10.1007/s11356-019-06959-1
Wan, W., Pepping, T. J., Banerji, T., Chaudhari, S., & Giammar, D. E. (2011). Effects of water chemistry on arsenic removal from drinking water by electrocoagulation. Water Research, 45(1): 384–392. https://doi.org/10.1016/j.watres.2010.08.016
Weerasundara, L., Ok, Y.-S., & Bundschuh, J. (2021). Selective removal of arsenic in water: A critical review. Environmental Pollution, 268: 115668. https://doi.org/10.1016 /j.envpol.2020.115668
Yamamura, S., & Amachi, S. (2014). Microbiology of inorganic arsenic: From metabolism to bioremediation. Journal of Bioscience and Bioengineering, 118(1): 1–9. https://doi.org /10.1016/j.jbiosc.2013.12.011
Yamamura, S., Ike, M., & Fujita, M. (2003). Dissimilatory arsenate reduction by a facultative anaerobe, Bacillus sp. Strain SF-1. Journal of Bioscience and Bioengineering, 96(5): 454–460. https://doi.org/10.1016/S1389-1723(03)70131-5
Yang, H.-C., Fu, H.-L., Lin, Y.-F., & Rosen, B. P. (2012). Chapter Twelve—Pathways of Arsenic Uptake and Efflux. In: J. M. Argüello & S. Lutsenko (Eds.), Current Topics in Membranes (Vol. 69, pp. 325–358). Academic Press. https://doi.org/10.1016/B978-0-12-394390-3.00012-4
Yin, S., Zhang, X., Yin, H., & Zhang, X. (2022). Current knowledge on molecular mechanisms of microorganism-mediated bioremediation for arsenic contamination: A review. Microbiological Research, 258: 126990. https://doi.org/10.1016/ j.micres.2022.126990