Dibya Sree Dutta1& Pankaj Bharali1&2
1Biological Sciences and Technology Division, CSIR-North East Institute of Science and Technology, Jorhat 785006, Assam, India
2Academy of Scientific and Innovative Research (AcSIR), Ghaziabad 201002, India
*Corresponding author email: pbharali@neist.res.in/pankajbharali98@gmail.com
Abstract
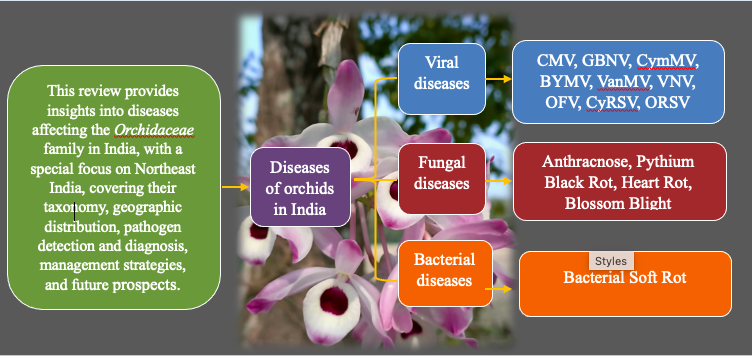
Orchids, a diverse and economically significant plant group, are highly susceptible to fungal, bacterial, and viral diseases, which adversely affect their cultivation and market value. Northeast India, renowned for its rich biodiversity, comprises the eight states of Arunachal Pradesh, Assam, Manipur, Meghalaya, Mizoram, Nagaland, Sikkim, and Tripura. This region serves as a hotspot for orchid diversity; however, its humid subtropical climate also fosters the proliferation of various plant diseases. Major fungal pathogens such as Colletotrichum, Fusarium, and Phytophthora, bacterial soft rot caused by Erwinia carotovora, and viral infections including CMV, ORSV, and CymMV lead to severe plant damage. Despite the widespread occurrence of these diseases, research on orchid pathology, particularly in Assam, remains limited, posing challenges to effective management. This review provides a comprehensive overview of the major orchid diseases in India, with a special emphasis on the northeastern region.
Keywords: Diseases, Northeast India, Orchids
GRAPHICAL ABSTRACT
Introduction
Orchids, classified under the family Orchidaceae, constitute one of the most diverse and widely distributed plant groups, comprising nearly 28,000 species and over 100,000 registered hybrids worldwide (Govaerts et al., 2017; Jangyukala and Hemanta, 2021). India hosts approximately 1,300 orchid species, with nearly 800 species found across the northeastern region, making it a significant biodiversity hotspot for these plants (Hinsley et al., 2018). The species distribution across the northeastern states includes Arunachal Pradesh (686), followed by Sikkim (501) and Assam (406), respectively. Globally, the orchid industry holds substantial economic value in floriculture, estimated at approximately $400 billion, with major producers including Thailand, Singapore, and Taiwan (DITP News, 2015).
Orchid cultivation, despite its economic value and global demand, is severely impacted by biotic and abiotic stresses, with diseases being a major constraint. Orchids are highly susceptible to fungal, bacterial, and viral infections, leading to reduced plant vigour, poor flower quality, and economic losses. Black rot is a major fungal threat, causing extensive tissue decay (Uchida, 1994; Erwin and Ribeiro, 1996). Other fungal diseases like anthracnose and blossom blight further affect production. Bacterial soft rot also causes severe damage to orchid crops (De et al., 2019).
In addition to fungal and bacterial infections, viruses pose a major challenge to orchid cultivation. Cymbidium Mosaic Virus (CymMV) and Odontoglossum Ringspot Virus (ORSV) are among the most common viruses affecting orchids, causing mosaic patterns, necrosis, and floral deformations, thereby reducing plant vigour and commercial value (Varma and Ramachandran, 1994; Sherpa et al., 2003; Bhat et al., 2006; Sherpa et al., 2006a and 2006b). Once infected, viral diseases are difficult to control, as there are no effective chemical treatments available, making prevention and quarantine measures essential. In India, at least 10 viruses have been reported on cultivated orchids (Table 1).
The northeastern region of India, with its humid subtropical climate and high rainfall, provides ideal conditions for the proliferation of pathogens, making orchids in this region particularly susceptible to various diseases. The lack of proper disease management strategies, along with limited awareness among growers, further exacerbates the problem. Therefore, understanding the pathology, distribution, and management of orchid diseases is crucial for sustainable orchid production and conservation. This review analyses major orchid diseases in India, focusing on the Northeast, highlighting symptoms and causal organisms to aid effective disease control for growers and researchers.
- Important viruses of orchids
1.1 Cucumber mosaic virus CMV
Cucumber Mosaic Virus (CMV), belonging to the Bromoviridae family and Cucumovirus genus, is a widely distributed pathogen affecting numerous plant species, including orchids. It was first identified in vanilla plants in French Polynesia, where it caused severe vine distortions (Farreyrol et al., 2001). Subsequent reports confirmed its presence in Réunion and India (Madhubala et al., 2005; Sharma et al., 2005).
The genome of CMV is tripartite and positive-sense RNA, encapsulated in isometric particles roughly 32 nm in diameter. Its genome encodes five key proteins, including two polymerase components, i.e., RNA1 and RNA2, the 2b protein involved in viral movement and suppression of host defences, a 30K movement protein (MP), and a coat protein (CP) responsible for encapsidation. Some CMV strains also carry satellite RNAs (satRNAs), which can influence disease symptoms. With the ability to infect more than 1,000 plant species across 30 families and being transmitted in a non-persistent manner by over 80 aphid species, CMV is highly adaptable and poses a significant threat to global agriculture (Gallitelli, 2000).
1.2 Groundnut Bud Necrosis Virus
Groundnut Bud Necrosis Virus (GBNV), belonging to the Tospovirus genus, has been identified as a pathogen affecting Phalaenopsis orchids in Sikkim, India. Infected plants displayed symptoms such as mild chlorotic spots, dark-green patches with chlorotic margins and mosaic patterns. Transmission Electron Microscopy (TEM) analysis of symptomatic leaf samples confirmed the presence of tospovirus-like particles ranging from 80 to 110 nm in diameter. This study marked the first global report of GBNV infecting Phalaenopsis orchids, including in India (Pant et al., 2019).
1.3 Cymbidium ringspot virus
Cymbidium Ringspot Virus (CyRSV), first identified in Cymbidium orchids by Hollings and Stone in 1963, is classified under the Tombusviridae family and Tombusvirus genus, with Tomato Bushy Stunt Virus as its type species. This virus shares serological similarities with an isometric virus found in Trifolium repens (white clover) and was initially reported from southern England. Infected Cymbidium orchids typically develop chlorotic ring mottle and exhibit stunted growth, though these symptoms are not exclusively linked to CyRSV. The virus spreads through mechanical transmission and can infect over 60 plant species. Unlike many plant viruses, CyRSV is not transmitted via seeds or insect vectors but remains highly stable in the environment.
In India, Cymbidium Ringspot Virus (CyRSV) has been detected in Cymbidium elegans and Cymbidium longifolium (Sharma et al., 2005). Structurally, CyRSV is a non-enveloped, icosahedral virus with a spherical shape and T=3 symmetry, measuring approximately 28–30 nm in diameter. Its genome comprises a single-stranded, positive-sense RNA (ssRNA) spanning 4,733 nucleotides and contains five major open reading frames (ORFs). The 33 kDa and 92 kDa proteins play a role in viral replication, the 41 kDa protein facilitates movement of virus within the host, while the 22 kDa and 19 kDa proteins contribute to capsid formation. Additionally, a small 4 kDa protein (ORF6) has been identified in the 3′ non-coding region through computational analysis (Boyko and Karasev, 1992). The stability and wide host range of CyRSV makes it a significant pathogen affecting ornamental orchids (Hollings and Stone, 1963; Boyko and Karasev, 1992; Sharma et al., 2005).
1.4 Potyviruses
Potyviruses constitute a significant group of plant viruses that impact a diverse range of crops, including orchids. In India, the Bean yellow mosaic virus (BYMV) has been identified in orchids such as vanilla, leading to symptoms such as mosaic and leaf malformations (Bhai et al., 2003; Bhat et al., 2004). Vanilla mosaic virus (VanMV), initially discovered in Vanilla tahitensis, shares a close genetic relationship with Dasheen mosaic virus (DsMV) and is spread by the aphid Myzus persicae (Farreyrol et al., 2006). Vanilla necrosis virus (VNV), first recorded in Tonga, was later classified as a strain of Watermelon mosaic virus (WMV-Tonga) (Pearson and Pone, 1988). Additionally, Calanthe mild mosaic virus (CalMMV) has been detected in orchids, causing mosaic and necrotic symptoms. Genomic analysis of VanMV has revealed a polyprotein structure characteristic of Potyviruses, with phylogenetic studies showing its close evolutionary link to South Pacific strains of DsMV (Puli’uvea et al., 2017).
1.5 Orchid fleck virus
Orchid fleck virus (OFV) was initially detected in Cymbidium orchids in Japan, where it was associated with chlorotic and necrotic flecks on leaves (Doi et al., 1977). Since then, the virus has been documented in multiple countries, infecting a range of orchid species and leading to floral deformities (Blanchfield et al., 2001). OFV is transmitted persistently by Brevipalpus mites (Kondo et al., 2003) and belongs to the proposed genus Dichorhabdovirus within the Rhabdoviridae family (Dietzgen et al., 2014). Morphologically, the virus has a bacilliform shape and possesses a (bipartite) negative-sense RNA genome. In India, virus particles resembling OFV have been observed in Coelogyne elata, C. flaccida, and several Cymbidium species found in Sikkim and Darjeeling (Pant, 2008). Symptoms in infected plants include chlorotic flecks, necrotic rings, and yellow spotting.
1.6 Odontoglossum ringspot virus
Odontoglossum ringspot virus (ORSV), a Tobamovirus from the family Virgaviridae, was first identified in Odontoglossum grande in the United States (Jenson and Gold, 1951). It later spread to Cymbidium, Cattleya, and other orchids, causing symptoms such as ringspots, chlorotic streaks, mosaicism, necrosis, and flower variegation (Lawson and Hsu, 1995). In India, the first report of ORSV was on Cymbidium species (Sherpa et al., 2004, 2006a, 2006b). It commonly co-infects orchids with Cymbidium mosaic virus (CymMV), leading to severe symptoms like necrosis and leaf cracking (Navalinskiene et al., 2005).
The virus is characterized by rod-shaped particles measuring 300–318 nm in length and possesses a positive-sense single-stranded RNA genome of approximately 6609 nucleotides (Chang et al., 1996). It encodes four proteins: RNA-dependent RNA polymerase (126/183 kDa), movement protein (33 kDa), and coat protein (18 kDa). ORSV spreads mechanically through contaminated tools and plant contact, with no known insect vectors. Studies indicate low genetic variability among global isolates, suggesting its stable genomic structure (Rao et al., 2015).
Table 1. List of orchid viruses reported across India and Northeast India
| Sl. No. | Family | Genus | Genome | Virus name | Distribution | References |
| 1 | Bromoviridae | Cucumovirus | RNA | Cucumber mosaic virus (CMV) | Kerala, Sikkim Karnataka, | Bhat et al. (2004), Sharma et al (2005) |
| 2 | Bunyaviridae | Tospovirus | RNA | Groundnut bud necrosis virus (GBNV) | Sikkim | Pant et al. (2019) |
| 3 | Flexiviridae | Potexvirus | RNA | Cymbidium mosaic virus (CymMV) | Sikkim, Darjeeling hills Karnataka and Kerala | Varma and Ramachandran (1994), Sherpa et al. (2003), Bhat et al. (2006) |
| 4 | Potyviridae | Potyvirus | RNA | Bean yellow mosaic virus (BYMV) | Kerala, Northeastern states | Sharma et al. (2005) |
| 5 | Potyviridae | Potyvirus | RNA | Vanilla mosaic virus (VanMV) | Kerala and Karnataka | Bhat et al. (2004) |
| 6 | Potyviridae | Potyvirus | RNA | Vanilla necrosis virus (VNV) | Kerala and Karnataka | Bhat et al. (2004) |
| 7 | Potyviridae | Potyvirus | RNA | Calanthe mild mosaic virus (CalMMV) | Sikkim | Singh et al. (2007); Pant et al. (2017) |
| 8 | Rhabdoviridae | Rhabdovirus | RNA | Orchid fleck virus (OFV) | Sikkim and Darjeeling | Pant (2008) |
| 9 | Tombusviridae | Tombusvirus | RNA | Cymbidium ring spot virus (CyRSV) | Sikkim | Sharma et al. (2005) |
| 10 | Virgaviridae | Tobamovirus | RNA | Odontoglossum ring spot virus (ORSV) | Sikkim, Darjeeling hills | Sherpa et al. (2006a, 2006b) |
- Fungal diseases of orchids: Several fungal pathogens infect orchids, leading to symptoms such as leaf spots, rots, and blights. The most common fungal diseases include:
2.1 Anthracnose
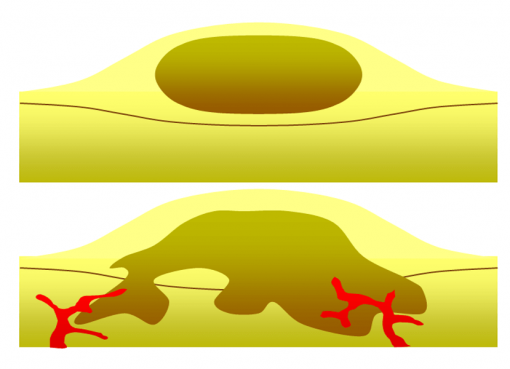
Anthracnose, caused by Colletotrichum gloeosporioides and C. orchidacearum, is a common fungal disease in orchids, characterized by sunken, oval lesions that range from reddish-brown to dark brown or grey. These lesions often appear on leaves, significantly affecting the plant’s aesthetic value and overall health. The disease thrives in warm, humid conditions, making orchids in greenhouse environments particularly susceptible. Effective management of anthracnose involves removing infected plant parts and enhancing air circulation to minimize moisture levels, which helps prevent the disease from spreading. Regular fungicidal applications of Carbendazim (Bavistin) at 1 g/L at 10-day intervals have been found effective in controlling the disease and preventing its spread (De et al., 2019).
2.2 Pythium Black Rot
Pythium Black Rot, caused by Pythium ultimum, is a destructive fungal disease in orchids, primarily affecting seedlings in humid conditions. The disease manifests as blackened leaves that eventually fall off, leading to severe plant deterioration. High moisture levels and poor drainage contribute to its rapid spread. Effective anthracnose management includes removing and destroying infected leaves, reducing irrigation, and relocating plants to a drier environment to limit fungal growth. Additionally, chemical control through fungicidal sprays of Metalaxyl (0.1%) or Mancozeb (0.2%) has been proven effective in managing the disease and preventing further infection (De et al., 2019).
2.3 Heart Rot
Heart Rot, caused by Phytophthora palmivora, is a severe fungal disease affecting orchids, particularly Cattleya, Phalaenopsis, and Vanda species. The disease is characterized by yellowing of leaves, premature foliage drop, and rotting of pseudobulbs, leading to significant plant decline. High humidity and excessive moisture favour its development, making well-drained growing conditions essential for prevention. Management strategies include the prompt removal and destruction of infected plants (rouging) to prevent disease spread. Additionally, the application of Metalaxyl (0.1%) has been found effective in controlling the pathogen and limiting further infection (De et al., 2019).
2.4 Blossom Blight
Blossom Blight, caused by Botrytis cinerea, is a common fungal disease affecting orchids, particularly during cool and humid conditions. The disease manifests as numerous dark spots on flower petals, which may enlarge and create a shot-hole effect, reducing the aesthetic value of the blooms. Older flowers are more susceptible to infection, and high moisture levels exacerbate disease progression. Effective management includes maintaining proper air circulation and reducing excessive humidity. Chemical control measures such as spraying Bavistin at 1 g/L or Indofil Z at 2 g/L at 7-day intervals have been found to effectively control the disease and prevent further spread (De et al., 2019).
- Bacterial Diseases of Orchids: Bacterial infections in orchids often cause soft rots, affecting the pseudobulbs and leaves. The most prominent bacterial disease is:
3.1 Bacterial Soft Rot
Erwinia carotovora is the causal agent of bacterial soft rot, a highly destructive disease that affects orchids, leading to rapid tissue decay and plant deterioration. The infection begins as small, circular, blister-like dark green spots on the leaves, which rapidly progress into a soft, pulpy rot with a foul odour. High humidity and poor air circulation favour disease development, leading to severe tissue breakdown in the pseudobulbs. To manage the disease, infected plant parts should be removed promptly, and environmental conditions should be adjusted to reduce excess moisture. Chemical control through the application of Streptocycline (0.1%) has been found effective in suppressing bacterial growth and preventing further spread (De et al., 2019).
Conclusion
Orchids are among the most economically and ecologically significant flowering plants, with India, particularly the northeastern region, serving as a crucial biodiversity hotspot. Despite their ornamental and commercial value, orchid cultivation faces severe challenges due to various fungal, bacterial, and viral diseases that impact plant health, flower quality, and overall marketability. While numerous studies have documented orchid diseases globally, research efforts in India, especially in Assam, remain limited. A significant research gap exists in Assam, where no comprehensive studies have been conducted on the pathological aspects of orchids. Although diseases affecting orchids have been frequently observed in the region, systematic investigations into their causal agents, epidemiology, and management strategies remain scarce. This lack of detailed research hinders the development of effective disease control measures, leaving orchid growers vulnerable to recurring infections and potential economic losses. Addressing this research gap through systematic investigations and disease management studies is essential for ensuring the sustainable growth of the orchid industry in India.
References
Bhai, R.S., Thomas, J., Potty, S.N., Geetha, L. and Solomon, J.J. (2003). Mosaic disease of vanilla (Vanilla plantifolia Andrews) – the first report from India. Journal of Spices and Aromatic Crops, 12: 80–82.
Bhat, A.I., Bhadramurthy, V., Saju, S. and Hareesh, P.S. (2006). Detection and identification of Cymbidium mosaic virus infecting vanilla (Vanilla plantifolia Andrews) in India based on coat protein gene sequence relationships. Journal of Plant Biochemistry and Biotechnology, 15: 33–37.
Bhat, A.I., Venugopal, M.N., Pant, R.P. and Bhai, R.S. (2004). Occurrence and distribution of viral diseases of vanilla (Vanilla plantifolia Andrews) in India. Journal of Spices and Aromatic Crops, 13: 143–148.
Blanchfield, A.L., Mackenzie, A.M., Gibbs, A., Kondo, H., Tamada, T. and Wilson, C.R. (2001). Identification of Orchid fleck virus by reverse transcriptase polymerase chain reaction and analysis of isolate relationships. Journal of Phytopathology, 149: 713–718.
Boyko, V.P. and Karasev, A.V. (1992). Tombusvirus genome may encode the sixth small protein near its 3’ terminus. Virus Genes, 6: 143–148.
Chang, C.C., Wong, S.M., Mahtani, P.H., Loh, C.S., Goh, C.J., Kao, M.C.C., et al. (1996). The complete sequence of a Singapore isolate of Odontoglossum ringspot virus and comparison with other tobamoviruses. Gene, 171: 155–161.
De, T., De, L.C., Singh, D.R., Roy, A. and Chowdhury, A.K. (2019). Diseases in orchids [Poster]. North Bengal Agricultural University & ICAR-NRC for Orchids. Available at: https://www.researchgate.net/publication/338208475.
Dietzgen, R.G., Kuhn, J.H., Clawson, A.N., Freitas-Astúa, J., Goodin, M.M., Kitajima, E.W., et al. (2014). Dichorhavirus: a proposed new genus for Brevipalpus mite-transmitted, nuclear, bacilliform, bipartite, negative-strand RNA plant viruses. Archives of Virology, 159: 607–619.
DITP News (2015). Tropical Thai orchids are in high demand around the world. Thai Trade Center Los Angeles. Available at: www.thailandhorizon.com.
Doi, Y., Chang, M.U. and Yora, K. (1977). Orchid fleck virus. CMI/AAB Descriptions of Plant Viruses (183).
Erwin, D.C. and Ribeiro, O.K. (1996). Phytophthora diseases worldwide. American Phytopathological Society, St. Paul, MN.
Farreyrol, K., Pearson, M.N., Grisoni, M., Cohen, D. and Beck, D.L. (2006). Vanilla mosaic virus isolates from French Polynesia and the Cook Islands are Dasheen mosaic virus strains that exclusively infect vanilla. Archives of Virology, 151: 905–919.
Gallitelli, D. (2000). The ecology of Cucumber mosaic virus and sustainable agriculture. Virus Research, 71: 9–21.
Govaerts, R., Bernet, P., Kratochvil, K., Gerlach, G., Carr, G., Alrich, P., et al. (2017). World checklist of Orchidaceae. Kew: Facilitated by the Royal Botanic Gardens. Available at: http://apps.kew.org/wcsp (Accessed March 23, 2017).
Hinsley, A., De Boer, H.J., Fay, M.F., LM, G.S.W.G., Gunasekara, R.S., Kumar, P., et al. (2018). A review of the trade in orchids, and its implications for conservation. Botanical Journal of the Linnean Society, 186: 435–455. doi: 10.1093/botlinnean/box083.
Hollings, M. and Stone, O.M. (1963). Cymbidium ringspot (a previously undescribed virus). Glasshouse Crop Research Institute Annual Report, p. 146.
Jangyukala, M. and Hemanta, L. (2021). Review on advances in production of hybrids in orchids. Journal of Crop Science and Technology, 10: 15–20. doi: 10.37591/RRJoCST.
Kondo, H., Maeda, T. and Tamada, T. (2003). Orchid fleck virus: Brevipalpus californicus mite transmission, biological properties, and genome structure. Experimental and Applied Acarology, 30: 215–223.
Lawson, R.H. and Hsu, H.T. (1995). Orchid. In: Loebenstein, G., Lawson, R.H., Brunt, A.A. (Eds.), Virus and Virus-Like Diseases of Bulb and Flower Crops. John Wiley & Sons, pp. 409–420.
Madhubala, R., Bhadramurthy, V., Bhat, A.I., Hareesh, P.S., Retheesh, S.T. and Bhai, R.S. (2005). Occurrence of Cucumber mosaic virus on vanilla (Vanilla plantifolia Andrews) in India. Journal of Biosciences, 30: 339–350.
Navalinskiene, M., Rougalas, J. and Samuitiene, M. (2005). Viral diseases of flower plants 16. Identification of viruses affecting orchids (Cymbidium Sw.). Biologia (Lahore, Pakistan), 2: 29–34.
Pant, R.P. (2008). Association of Rhabdovirus-like particles from Coelogyne species from Sikkim and Kalimpong. Annual Report, National Research Centre for Orchids, Pakyong, Sikkim, India, p. 28.
Pant, R.P., Basavaraj, Y.B., Srivastava, N., Bhattarai, A., Kumar, A., Baranwal, V.K., et al. (2019). First report of Groundnut bud necrosis virus infecting Phalaenopsis in India. New Disease Reports, 39: 17.
Singh, M.K., Sherpa, A.R., Hallan, V. and Zaidi, A.A. (2007). A potyvirus in Cymbidium s in Northern India. Australasian Plant Disease Notes, 2(1): 11–13.
Uchida, J.Y. (1994). Diseases of orchids in Hawaii. Plant Disease, 78: 220–224. doi: 10.1094/PD-78-0220.
Varma, A. and Ramachandran, P. (1994). Viral diseases of orchids. Journal of Orchid Society of India, 8: 15–18.